GutAlive®: Anaerobic Kit for Microbiome Collection that Preserves Bacterial Composition and Viability
GutAlive® is a revolutionary microbiome collection kit specifically designed to ensure the preservation of the composition and bacterial diversity of fecal samples. By creating an anaerobic atmosphere, this leading-edge device enables the viability of bacterial species without altering their original diversity.
At Microviable Therapeutics, we specialize in biotherapeutics development and innovate in the field of microbiota-based therapies. In line with this, we have developed a system that allows us to collect and transport intestinal microbiota while maintaining the viability and diversity of microorganisms for research purposes.
GutAlive guarantees anaerobic conditions that allow the preservation of bacterial diversity
A major challenge in fecal sample collection has always been the loss of microorganisms during the collection and shipping process.
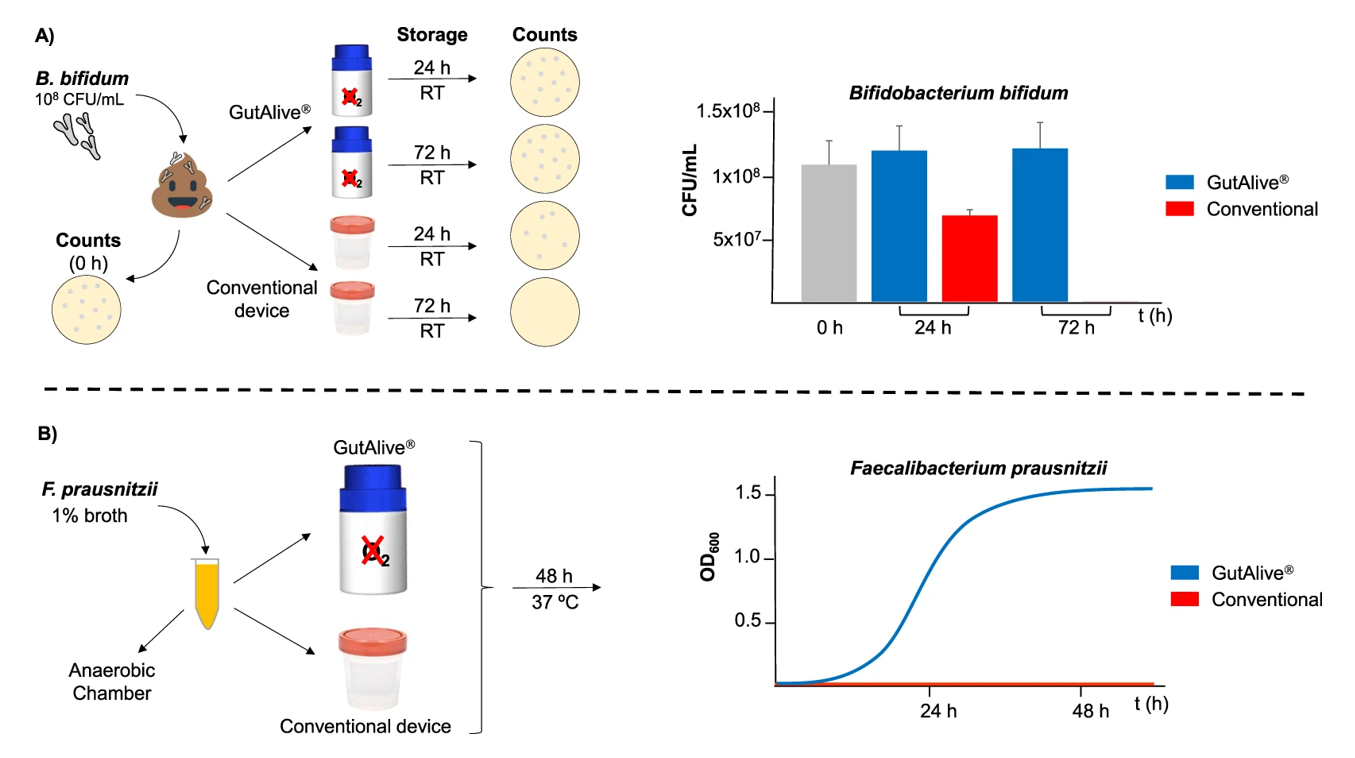
Donor manipulation, as well as preservation temperature, affect the integrity of microbial populations present in the feces. The deficiencies in packaging closure and the time elapsed between collection and microbiota analysis in the laboratory also impact them. The GutAlive microbiome collection kit provides a completely effective isolation that fully protects the microbial diversity of the sample.
GutAlive® is a unique device capable of stabilizing the specimen, ensuring the survival of cells. Even in the case of strict anaerobic microorganisms (EOS), it manages to preserve their original composition by creating an oxygen-free atmosphere. Compared to other containers in its class, GutAlive® is also an excellent option for preserving DNA and conducting NGS technologies and metagenomic sequencing.
The GutAlive anaerobic microbiome collection kit guarantees complete isolation of the fecal sample at room temperature to protect microbial diversity.

Advantages and Standardization of Protocols
The GutAlive® microbiome collection kit revolutionizes fecal sampling processes. It allows for the standardization of protocols for the collection and shipment of fecal samples. This technical advancement provides a significant advantage for the development of scientific studies and the analysis process in the clinical and research fields.
One notable feature of GutAlive® is its ability to collect a fecal sample with a large volume of up to 120 grams. A single sample enables various applications, analyses, and microorganism isolation procedures, as well as clinical use for fecal microbiota transplantation.
Another significant advantage is its ability to ensure sample stability for 72 hours without refrigeration. This ensures suitable conditions for sample collection and transportation to the laboratory while avoiding additional costs.
Compared to conventional devices, the GutAlive® anaerobic fecal collection kit represents a considerable advancement that completely changes the landscape of microbiota research. Its effectiveness and practicality make it an essential tool for standardization and improvement of studies that require fecal sample collection in various contexts.
GutAlive® is a refrigeration-free microbiome collection kit that allows for the collection of fecal samples up to 120 grams and ensures their stability for 72 hours.
Advantages and Standardization of Protocols
The GutAlive® microbiome collection kit revolutionizes fecal sampling processes. It allows for the standardization of protocols for the collection and shipment of fecal samples. This technical advancement provides a significant advantage for the development of scientific studies and the analysis process in the clinical and research fields.
One notable feature of GutAlive® is its ability to collect a fecal sample with a large volume of up to 120 grams. A single sample enables various applications, analyses, and microorganism isolation procedures, as well as clinical use for fecal microbiota transplantation.
Another significant advantage is its ability to ensure sample stability for 72 hours without refrigeration. This ensures suitable conditions for sample collection and transportation to the laboratory while avoiding additional costs.
Compared to conventional devices, the GutAlive® anaerobic fecal collection kit represents a considerable advancement that completely changes the landscape of microbiota research. Its effectiveness and practicality make it an essential tool for standardization and improvement of studies that require fecal sample collection in various contexts.
GutAlive® is a refrigeration-free microbiome collection kit that allows for the collection of fecal samples up to 120 grams and ensures their stability for 72 hours.

Microbiome collection kit for the exclusive use of researchers and clinician
Exclusively intended for professional use, the GutAlive® microbiome collection kit is authorized as a medical device with License 7245-PS. Only research centers and specialized clinics have access to this device.
Currently, its application is being standardized in more than 20 countries. Numerous hospital centers, R&D companies, and medical research entities worldwide have already included the use of GutAlive® in their analysis procedures. Its excellent quality and anaerobic capacity prevent changes in microbial dynamics and maintain the sample’s conditions unaltered. Its increasingly widespread use has turned this microbiome collection device into an indispensable tool.
The Spanish Gastroenterology Association has recognized the importance of incorporating GutAlive® into its protocols for Fecal Microbiota Transplantation. This innovative solution is endorsed by hundreds of experts as the best alternative for preserving the original biodiversity of samples and ensuring consistent results.
Microviable Therapeutics revolutionizes the way microbiota samples are obtained for study. We guarantee precise collection that preserves the integrity of the sample. GutAlive® is positioned as the most effective microbiome collection kit in the world.
See More Details about GutAlive® Stool Collection Kit